Products
Host Cell Residue Detection
Process Impurity Assay
Advnetitious Agent/Microorganism Testing
Pyrogen / Endotoxin Detection
Genetic Stability
Cell Line Characterization
Viral Titer Assay
Detection Equipment
Biochemical Reagents
Recently, GeneSail Biotech (Shanghai) Co., Ltd. (hereinafter referred to as GeneSail Biotech) has successfully obtained the Class 1 New Drug Clinical Trial Approval issued by the National Medical Products Administration (NMPA) for its independently developed innovative drug GO321 Recombinant Oncolytic Vaccinia Virus Injection. This milestone signifies that GeneSail Biotech has stepped into the multi-product clinical research phase in the innovative R&D and industrialization of oncolytic vaccinia virus therapeutics.
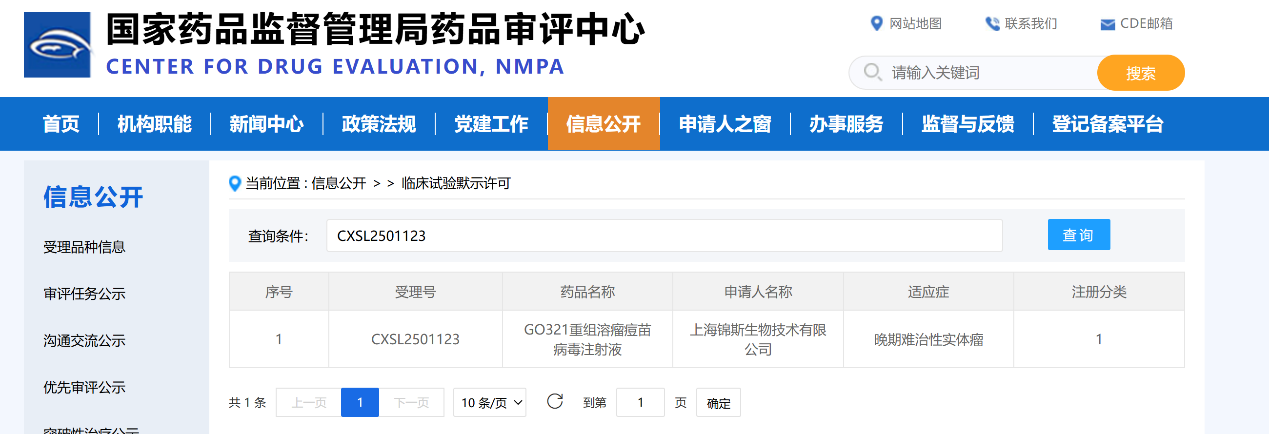
In this project, Huzhou Shenke Biotechnology Co., Ltd. (hereinafter referred to as HZSKBIOⓇ) leveraged its professional, fully validated NGS-based adventitious viral factor detection platform, and provided viral safety testing services for the client’s product with stringent high-standard protocols, fully meeting domestic and international regulatory requirements.
Going forward, driven by technological innovation and oriented toward high-quality services, HZSKBIOⓇ will continue to deepen the exploration and practice of new technologies for biopharmaceutical quality analysis. We will join hands with more industry partners to safeguard the high-quality development of China’s biopharmaceutical sector.
About GeneSail Biotech
GeneSail Biotech (Shanghai) Co., Ltd. dedicates itself to the innovative R&D and industrialization of oncolytic virus drugs. It owns an internationally leading technical platform for viral vector R&D and industrialization, enabling the development and production of various viral vector drugs complying with domestic and international regulatory requirements under GMP-compliant standards.
The core team of GeneSail Biotech has accumulated nearly 30 years of profound experience in innovative design of oncolytic virus drugs, preclinical pharmacological research, clinical research, pilot scale-up and commercial production process development, quality control, quality assurance and GMP manufacturing. Relying on its self-built technical platform, the company has developed a portfolio of innovative oncolytic virus pipelines with complete independent intellectual property rights. Up to now, two oncolytic virus drug candidates of GeneSail Biotech have been approved for clinical trials by the Center for Drug Evaluation (CDE) of the NMPA.
The company has received financial support from multiple science and technology programs, including the Innovation R&D Project of Shanghai Municipal Science and Technology Commission, the Gene Therapy Technology Platform Project of Shanghai Municipal Development and Reform Commission, and the Key R&D Program of the Ministry of Science and Technology of China. It has also been honored as a Shanghai "Specialized, Refined, Characteristic & Innovative" Enterprise.
About HZSKBIOⓇ
Founded in 2012, Huzhou Shenke Biotechnology Co., Ltd. is a National High-tech Enterprise and a national-level Specialized, Refined, Characteristic & Innovative "Little Giant" Enterprise certified by the Ministry of Industry and Information Technology.
The company focuses on the R&D and industrialization of pharmaceutical quality control technologies. It provides standardized testing reagents, professional testing equipment and technical services covering host residual nucleic acid/protein, exogenous risk factors, process residues, genetic stability and other testing items for global biopharmaceutical enterprises, CRO/CDMO organizations, scientific research institutions and regulatory authorities.
HZSKBIOⓇ has obtained ISO13485 certification and CNAS laboratory accreditation. Its core products have completed FDA DMF filing, supporting IND and BLA applications for pharmaceutical enterprises at home and abroad. With "standardized + customized" integrated system solutions, the company serves customers in multiple fields, stabilizes the biopharmaceutical supply chain, and empowers high-quality development of the industry.