Products
Host Cell Residue Detection
Process Impurity Assay
Advnetitious Agent/Microorganism Testing
Pyrogen / Endotoxin Detection
Genetic Stability
Cell Line Characterization
Viral Titer Assay
Detection Equipment
Biochemical Reagents
Process-Specific HCP Antibody & ELISA Kit for Global Regulatory Submission
Coverage >80% | Sensitivity ≤3 ng/mL | FDA/EMA/ICH Compliant
Host Cell Proteins (HCPs) are process-related impurities derived from the host organism used in the production of recombinant biopharmaceuticals, such as bacteria, yeast, mammalian, insect, or plant cells. During manufacturing, these heterogeneous protein impurities are inevitably introduced into the final product and must be strictly monitored throughout the production process and in the drug substance, as they pose potential immunogenicity risks and may impact product stability.
HCP residual levels are classified as a Critical Quality Attribute (CQA), requiring rigorous control. Generic HCP detection kits often fail to fully cover the specific host cell proteins present in individual manufacturing processes, as HCP profiles vary significantly across different host types and culture conditions. Therefore, utilizing process-specific anti-HCP antibodies and detection kits matched to your production cell line or strain is essential for accurate monitoring and regulatory compliance.
Our HCP Custom Antibody Development Services encompass the complete workflow from HCP antigen preparation, antibody generation, antibody coverage analysis, and sandwich ELISA method development to kit manufacturing (if required). Leveraging mature technical platforms and extensive project experience, we deliver precise, reliable, and process-specific residual protein monitoring solutions to ensure drug safety and quality control.
Key Risk: Incomplete Coverage — Generic HCP ELISA kits may lack antibodies against process-specific HCPs, leading to underestimated contamination levels and potential patient safety risks.
Key Challenge: Regulatory Compliance — Regulatory agencies require demonstration that HCP antibodies provide adequate coverage of the specific HCP population present in your manufacturing process, supported by comprehensive validation data.
| Regulatory Body | Requirement |
| FDA (U.S.) | The U.S. Food and Drug Administration requires that HCP detection methods used for product release demonstrate adequate antibody coverage and specificity. Process-specific or platform-specific HCP ELISAs are typically required for late-stage clinical trials and commercial products. |
| EMA (Europe) | The European Medicines Agency mandates that HCP assays be validated with appropriate coverage analysis to ensure detection of the majority of potential impurities. Orthogonal methods (e.g., mass spectrometry) are recommended to complement ELISA limitations. |
| NMPA (China) | The Chinese Pharmacopoeia and NMPA guidelines emphasize the importance of HCP monitoring as a CQA, requiring validated methods with demonstrated antibody coverage suitable for the specific manufacturing process. |
From Antigen Preparation to Regulatory Submission
We offer end-to-end HCP antibody development services tailored to your product's unique host cell system and manufacturing process. Our approach combines proprietary immunomagnetic bead separation (IMBS) technology, orthogonal coverage analysis, and multi-platform detection capabilities to deliver robust, release-ready methods and regulatory-compliant documentation.
Step 1: HCP Antigen Preparation & Characterization
We perform antigen representativeness analysis and prepare graded HCP antigens from your specific host cell system. Mock cell lysates or early process samples are characterized to ensure the immunogen accurately represents the HCP population present in your manufacturing process. Reference standards and calibrators are prepared and fully characterized.
Step 2: Animal Immunization & Antibody Generation
Using tiered immunization strategies tailored to different antigen groups, we generate high-quality polyclonal antibodies. The immunization process is closely monitored to maximize antibody coverage, specificity, and titer.
Step 3: Antibody Purification & Coverage Analysis
Antibodies are purified and evaluated for performance. Antibody combinations are screened to identify optimal pairs for sandwich ELISA. Coverage is assessed using proprietary IMBS-2D and IMBS-LC-MS orthogonal methods to ensure broad HCP recognition.
Step 4: ELISA Method Development & Validation
We develop and optimize sandwich ELISA methods specifically for your HCP profile. The method undergoes comprehensive performance validation including specificity, sensitivity, accuracy, precision, and stability studies according to compendial requirements (USP, EP, ChP).
Step 5: Kit Manufacturing & Regulatory Support
We deliver fully documented SOPs ready for immediate QC implementation. Customized ELISA kits can be manufactured under ISO 13485 quality management system. Comprehensive study reports and technical documentation are provided to support IND, BLA, MAA, and other regulatory submissions.
Our laboratory employs a comprehensive suite of HCP detection and analysis platforms to ensure method suitability across diverse biologic matrices:
Sandwich ELISA — Gold standard for HCP quantification with high sensitivity and throughput, customized to your specific process
IMBS-2D Coverage Analysis — Proprietary immunomagnetic bead separation combined with 2D gel electrophoresis for visual assessment of antibody coverage
IMBS-LC-MS Coverage Analysis— Orthogonal mass spectrometry-based method providing identification and quantification of individual HCPs recognized by the antibody, with higher sensitivity and resolution than traditional 2D-WB methods
LC-MS HCP Identification—Direct identification and quantification of individual high-risk HCPs in drug substance and process intermediates
All methods are paired with optimized sample preparation protocols and proprietary IMBS technology to ensure accurate and comprehensive HCP monitoring.
Advantage | What It Means for You |
Accredited Platforms | CNAS-accredited laboratory; ISO 13485 quality management system; fully in-house platforms with no secondary outsourcing: Animal Immunization Platform, Coverage Assessment Platform (IMBS-2D / IMBS-LC-MS), Standardized Reagent/Kit Production Line. |
Regulatory Compliance | Development programs aligned with USP, EP, and ChP requirements; complete regulatory submission packages including process-specific HCP detection methods and comprehensive validation reports. |
Extensive Experience | Proven commercial kit development experience with 10+ generic HCP ELISA kits marketed; extensive custom antibody development experience covering leading domestic antibody, recombinant protein, and vaccine companies; project experience spanning CHO, E. coli, yeast, and special host systems (e.g., plant cells). |
Full Lifecycle Support | Customized kit production services; antibody purification and labeling services; critical material re-manufacturing services; stability studies; comprehensive technical support throughout the product lifecycle. |
The following representative projects have successfully supported client submissions to FDA and EMA:
| No. | Client Type | Product Type | Host Cell | Project Phase |
| 1 | Public Biopharma | Recombinant Protein | CHO | Post-marketing Change |
| 2 | Public Biopharma | Monoclonal Antibody (mAb) | CHO | Phase Ⅲ Clinical Trial |
| 3 | Large Pharma Subsidiary | — | Plant Cell | IND |
| 4 | Large Pharma Subsidiary | Recombinant Proteins | E. coli | Post-marketing Change |
| 5 | Public Biopharma | Monoclonal Antibody (mAb) | CHO | Phase Ⅲ Clinical Trial |
| 6 | Large Pharma Subsidiary | Cell Therapy | Murine Cell | IND |
| 7 | Public Biopharma | — | CHO | / |
| 8 | Large Pharma Subsidiary | Monoclonal Antibody (mAb) | CHO | Phase Ⅰ Clinical Trial |
| 9 | Large Pharma Subsidiary | Monoclonal Antibody (mAb) | CHO | Phase Ⅲ Clinical Trial |
*The above represents a partial case portfolio
1. Consultation & Requirements—Project background;Service requirements;Proposal confirmation
2. Immunogen Preparation
3. Animal Immunization
4. Antibody Purification & Screening
5. ELISA Method Development
6. Project Delivery
Fundamentals
Q1: What is the difference between generic HCP kits and process-specific HCP antibodies?
A: Generic HCP kits are designed to detect common HCPs from major expression systems (e.g., CHO, E. coli) and are suitable for early-stage development. However, they may lack antibodies against process-specific HCPs that arise from your unique cell line, culture conditions, and purification process. Process-specific antibodies are generated using antigens from your actual manufacturing process, ensuring broader coverage of relevant HCPs and reducing the risk of false-negative results.
Q2: Why do different HCP ELISA kits sometimes give significantly different results for the same sample?
A: This is a well-documented industry phenomenon. Different kits use different antibody reagents raised against different antigen preparations, leading to varying recognition of the heterogeneous HCP population. A kit may underreport HCPs if its antibodies fail to recognize process-specific HCPs that co-purify with your product. This is precisely why regulatory agencies now emphasize product-specific evaluation and why we recommend orthogonal LC-MS confirmation to identify individual high-risk HCPs that ELISA may miss.
Regulatory Compliance
Q3: What coverage level is considered acceptable for regulatory submission?
A: Regulatory agencies typically expect HCP antibody coverage of >70% as assessed by 2D or equivalent methods. Our IMBS-2D method routinely achieves >70% coverage, while our orthogonal IMBS-LC-MS method typically achieves >80% coverage, providing enhanced confidence in HCP detection.
Q4: At what development stage should we switch from a generic kit to a process-specific HCP assay?
A: Generic HCP kits provide maximum flexibility during early preclinical and clinical development when HCP populations are highly variable. However, they become less appropriate from Phase III onwards, when drug substance must meet commercial specifications. At this stage, a process-specific assay is typically required to adequately monitor the unique HCP population associated with your approved bioprocess. That said, generic kits can still be used for late-phase development if sufficient data demonstrates that the kit meets required performance and coverage criteria for your specific process.
Technical Methods
Q5: Which host cell systems do you support?
A: We have extensive experience with CHO cells, E. coli, yeast (including Pichia pastoris), and special host systems such as plant cells and murine cells. Our platforms can be adapted to virtually any host organism used in biopharmaceutical manufacturing.
Q6: How do you handle low-molecular-weight (LMW) HCPs that are typically less immunogenic and may be underrepresented in standard antibody preparations?
A: LMW HCPs are indeed a common challenge because standard immunization regimes often underrepresent them. Our counter-strategy includes fractionation of HCP antigens prior to immunization—both high- and low-molecular-weight fractions are used simultaneously to ensure balanced antibody generation.
Method Validation
Q7: What are the critical validation parameters for a process-specific HCP ELISA, and what acceptance criteria should we aim for?
A: Key validation parameters include: specificity (no cross-reactivity with drug substance), sensitivity (LOD/LOQ), precision (intra/inter-assay CVs typically <20%), accuracy (spike recovery within 100±30%), linearity, dilutional linearity (CV% <20% across all dilutions), and robustness. A good standard curve should have at least six points with a wide working range (e.g., 0–200 ng/mL). In addition, the coverage rate shall be greater than 70% when tested by IMBS‑2D, and greater than 80% when tested by IMBS‑LC‑MS.
Application Scenarios
Q8: Can we use the same HCP assay for both in-process monitoring and final product release?
A: Yes, provided the assay is validated for both purposes. However, the validation focus differs: for in-process samples, emphasis is on accuracy (spike recovery), dilution linearity, and precision. For final product release, you must additionally demonstrate specificity, quantitation limit, and—critically—that high drug substance concentrations do not mask HCP detection (drug interference). The acceptance criteria (e.g., ≤100 ng/mL early stage, ≤10 ng/mL late stage) must be justified based on clinical risk, dose, and route of administration.
Technical Comparison
Q9: What is the difference between IMBS-2D and IMBS-LC-MS coverage analysis?
A: IMBS-2D combines immunomagnetic bead separation with 2D gel electrophoresis, providing visual assessment of coverage through spot comparison. IMBS-LC-MS uses mass spectrometry to identify and quantify individual HCPs recognized by the antibody, offering higher sensitivity, better resolution, and objective protein identification without the limitations of spot counting or membrane transfer artifacts.
Commercialization Assurance
Q10: What happens if our commercial HCP kit is discontinued or the supplier stops production? How does your full lifecycle support mitigate this risk?
A: Critical material discontinuation is a major supply chain risk for commercial products. Our full lifecycle support includes key material re-manufacturing services and stability studies to ensure continuity. We maintain master cell banks and antibody archives, enabling re-production of identical antibody reagents with matched coverage profiles. For clients who developed process-specific assays with us, we provide long-term storage, periodic re-qualification, and seamless transition protocols—ensuring your validated method remains available for the entire product lifecycle, regardless of external supplier changes.
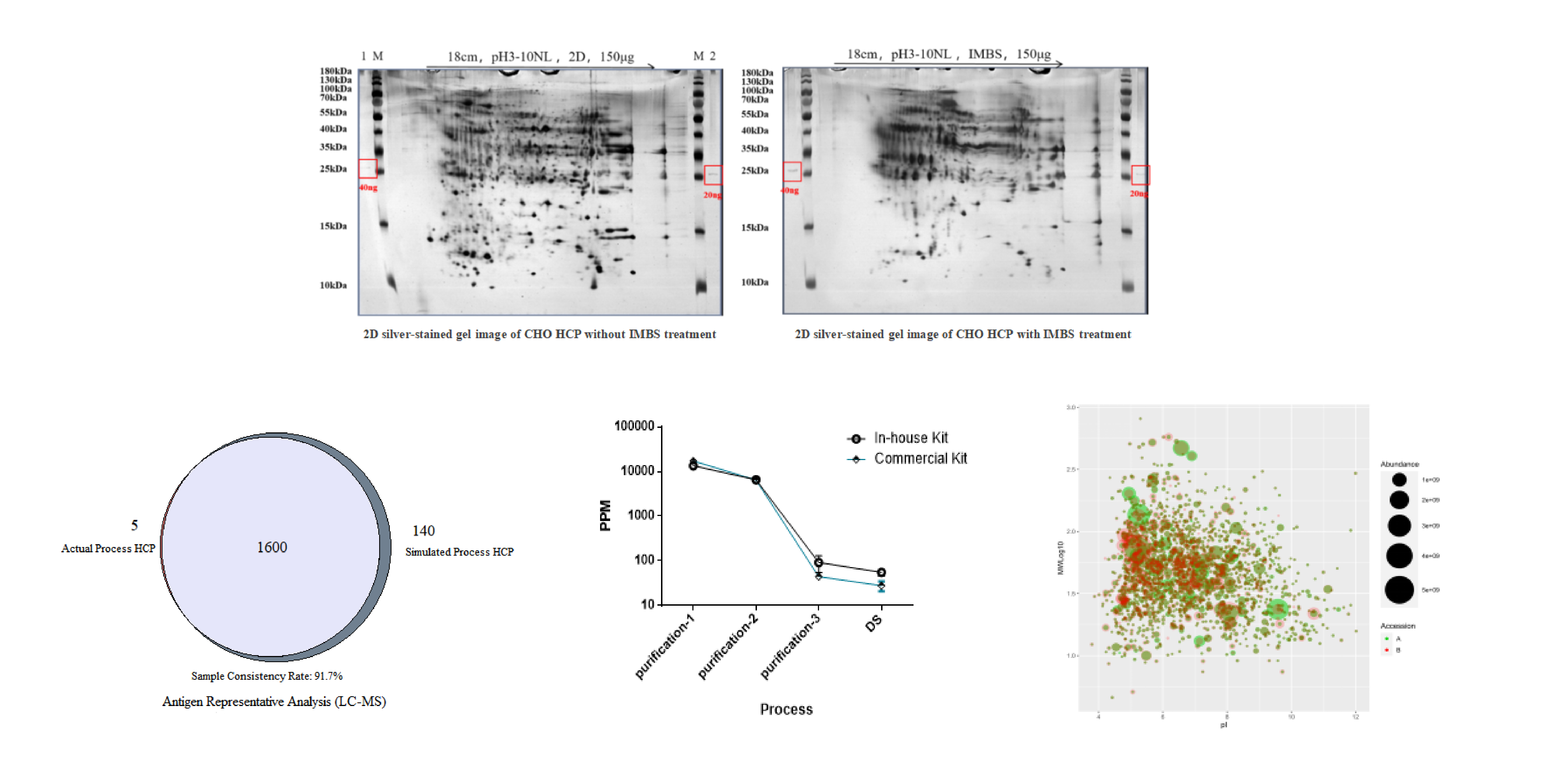
Project Background: A recombinant protein drug for dual U.S.-China submission required development of a process-specific CHO HCP detection kit.
Project Deliverables:
IMBS-2D coverage: >70%
IMBS-LC/MS coverage: ≥80%
Detection sensitivity: ≤3 ng/mL
Results: Successful bridging study with commercial kit to support method change
USP Chapter <1132>: Residual Host Cell Protein Measurement in Biopharmaceuticals
FDA Guidance for Industry: Q6B Specifications: Test Procedures and Acceptance Criteria for Biotechnological/Biological Products
EMA Guideline: Development, Production, and Quality Control of Medicinal Products Derived from Recombinant DNA Technology
ICH Q6B: Specifications: Test Procedures and Acceptance Criteria for Biotechnological/Biological Products
返回