Products
Host Cell Residue Detection
Process Impurity Assay
Advnetitious Agent/Microorganism Testing
Pyrogen / Endotoxin Detection
Genetic Stability
Cell Line Characterization
Viral Titer Assay
Detection Equipment
Biochemical Reagents
In recent years, recombinant collagen has been rapidly applied in medical dressings, wound repair, medical aesthetic injection, tissue engineering, regenerative medicine and other fields, with continuous expansion of industrial scale. According to the forecast data of Frost & Sullivan, the market size of domestic collagen API will reach 16 billion RMB by 2030, among which the market size of recombinant type III collagen raw materials will reach 10 billion RMB, accounting for over 60% of the overall market size of collagen raw materials.
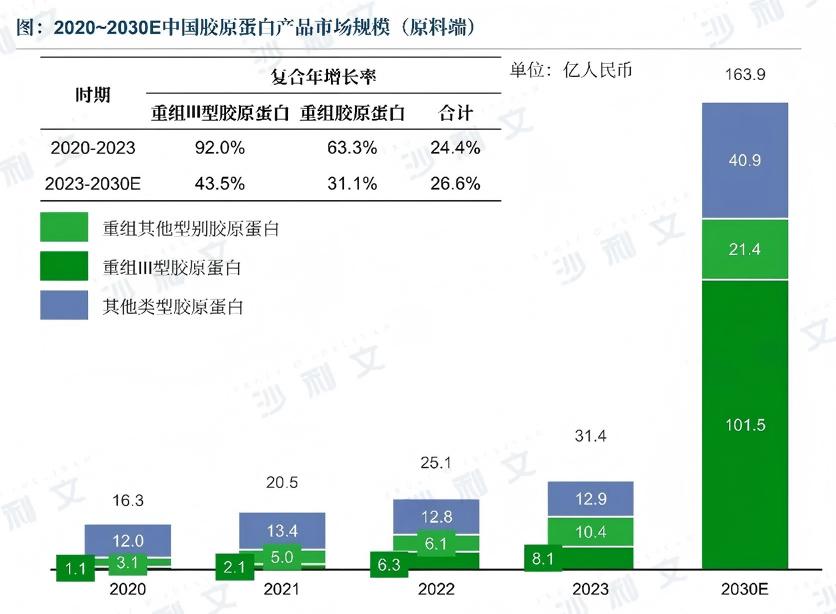
Source: Frost & Sullivan Global Recombinant Type III Collagen Market Research Report
With the rapid industrial development, the National Medical Products Administration (NMPA) has continuously tightened the review, approval and quality supervision. Host Cell DNA (HCD) is no longer only a key quality control item for Class III injectable medical devices. For recombinant collagen raw materials for medical devices produced by bioengineering expression systems, HCD serves as the core evaluation content for raw material quality control and registration filing regardless of whether the downstream finished products are Class II or Class III medical devices. It is a compliance and safety indicator that the industry must lay out in advance.
For enterprises engaged in the R&D and production of recombinant collagen, proactive layout of HCD detection and residue control is not only an inevitable compliance requirement, but also a key measure to be responsible for end users and maintain long-term product competitiveness.
01 What is Humanized Recombinant Collagen?
Collagen is the most abundant functional protein in mammals, accounting for 25% to 33% of total human protein, and widely distributed in skin, bone and tendon tissues. Collagen makes up 70% of the extracellular matrix (ECM) in the dermal layer of the skin, among which type I collagen playing a key skeletal supporting role accounts for as high as 80% to 90%. With aging, the triple helix structure of collagen is degraded and broken by metalloproteinases, directly leading to skin aging, decreased elasticity and wrinkle formation.
Commercial Classification of Collagen
Animal-derived collagen: such as bovine collagen and porcine collagen.
Humanized recombinant collagen: prepared by bioengineering technology, with a highly homologous sequence to natural human collagen and lower immunogenicity, becoming the mainstream of the market.
Regulatory Classification of Humanized Recombinant Collagen
Injection/implantation category: declared and regulated as Class III medical devices.
External dressing category:
· Class III medical devices: absorbable by the human body, used for chronic/deep wounds.
· Class II medical devices: non-absorbable, only for non-chronic superficial wound care.
Relying on the advantages of high biocompatibility and low immunogenicity, humanized recombinant collagen has expanded from facial anti-aging to tissue repair and regenerative medicine, including uterine cavity repair, post-mastectomy repair, wound healing and other scenarios. The wider the application scenarios, the higher the requirements for impurity residues and biosafety.
02 Why Pay Attention to HCD?
Humanized recombinant collagen is mostly produced by fermentation using expression systems such as Escherichia coli and Pichia pastoris. While engineering bacteria express target proteins, impurities such as host cell proteins and nucleic acids will inevitably remain. Host Cell DNA (HCD) is a key monitored safety risk substance with potential risks as follows:
· Tumorigenicity and genome integration risk: Residual DNA fragments carrying relevant sequences may insert into the human genome, posing potential hidden safety hazards.
· Immunogenicity risk: Exogenous DNA fragments can act as immunogens and trigger abnormal immune responses in the body, affecting product safety and clinical tolerance.
· Biosafety risk: Residual host cell-related sequences may bring potential biosafety risks.
03 Technical Path and Regulatory Standard System of HCD Detection
Standard Detection Technology: qPCR Method
Inclusion basis: Included in the Chinese Pharmacopoeia (2020 edition onwards), recognized by the industry and accepted by regulatory review authorities.
Detection principle: Design specific primers and probes targeting conserved repetitive sequences of the host genome for real-time quantification via fluorescence signals.
Core advantages: High sensitivity, strong specificity and good repeatability, enabling accurate detection of trace residues at fg level.
Application scenarios: Covering the whole process of R&D, pilot scale-up, large-scale production and registration filing.
Regulatory and Standard Requirements (Continuously Becoming Stringent)
Chinese Pharmacopoeia: HCD has been listed as a key impurity of biological products since the 2005 edition; the limit standards have been further refined since the 2010 edition. The residual amount of injectable products ≤10 ng/dose is an internationally accepted safety threshold.
YY/T 1849-2022 Recombinant Collagen: Applicable to recombinant collagen raw materials for medical devices, clearly defining HCD as a mandatory impurity control indicator for the first time.
YY/T 1888-2023 Humanized Recombinant Collagen: Further refining the requirements for HCD control, detection methods and safety evaluation.
Guidelines for the Evaluation of Humanized Recombinant Collagen Raw Materials (No.16, 2023, Medical Device Technical Evaluation Center of NMPA): It is recommended to refer to the limits of exogenous DNA residues of genetically engineered recombinant biological products expressed by E. coli or yeast in biological products.
YY/T 10001-2026 General Technical Requirements for Biotechnologically Derived Raw Materials Used in Cosmetics (implemented on May 1, 2027): If there are other risk substances in raw materials, risk information shall be truthfully disclosed, and safety assessment shall be carried out by combining multiple methods; reasonable limits for risk substances shall be formulated according to the assessment results to ensure that cosmetics produced under normal, reasonable and foreseeable dosage and use conditions comply with the Technical Specification for Cosmetic Safety.
The current regulatory logic is strict control over raw materials transmitted to finished products, and strict control over Class III medical devices transmitted to other categories.
As long as recombinant collagen raw materials are used for downstream finished products (whether Class II or Class III medical devices), HCD quality control shall be implemented in accordance with industry standards. With increasingly stringent review standards and on-site inspections, Class II manufacturers also need to incorporate HCD detection into the raw material quality control system. Proactive layout and compliance advance can reduce the risks of registration supplementary review and non-compliance.
Huzhou Shenke has built a qPCR detection platform covering multiple expression systems such as E. coli and Pichia pastoris. It provides one-stop solutions including methodological development, verification, routine detection, supporting detection reagents and equipment, supporting the whole-process quality control of enterprises from R&D to market release.
Itm No.e | Product Name | Specification/Model |
1610860 | Real-time Fluorescent PCR Detection System | SHENTEK-96S |
1609850 | rHCDpurify Pre-treatment System | SKRDP-32Pro |
SK030203D100 | Host Cell Residual DNA Sample Pretreatment Kit (Magnetic Bead Method) | 100 Extractions |
SK030205P100 | Pichia pastoris Residual DNA Detection Kit (PCR-Fluorescent Probe Method) | 100 Reactions |
1101107 | E.coli HCD Detection Kit (PCR-Fluorescent Probe Method) | 100 Reactions |
04 Data Analysis: Current Level of HCD Residue Control
As a supplier committed to quality and safety testing products and services for biopharmaceuticals, Huzhou Shenke has long focused on the quality control safety of upstream raw materials and terminal products in the medical aesthetics industry.
Based on statistical detection data of hundreds of batches of products commissioned by mainstream domestic collagen manufacturers in the past two years (covering recombinant collagen Type I, Type III and Type V, mainly based on Pichia pastoris expression system), the overall industry quality control level has been significantly improved:
✔ Leading enterprises have stably controlled HCD residues at pg level, far below regulatory limits.
✔ Over 80% of samples are below the kit limit of quantification (0.001 pg/μL).
✔ The purification process is continuously upgraded, and the impurity removal efficiency remains stable under large-scale production.
Driven by strict regulations and continuous upgrading of production processes, especially the improvement of chromatographic purification technology, the impurity removal efficiency remains stable even with expanded production scale, and the process purification capability of leading manufacturers has become highly mature.
HCD control capability has become the core symbol of enterprise process maturity, quality system and compliance competitiveness. Enterprises with listed products, under R&D, or deploying Class II and Class III medical device tracks have gradually included HCD as a mandatory testing item.
05 Enterprise Compliance Layout Suggestions: Advance Planning & Whole-process Quality Control
1. Advance planning at R&D stage: Design HCD control strategy synchronously during strain construction and expression system selection to reduce later transformation costs.
2. Continuous optimization at process stage: Verify impurity removal efficiency by combining chromatographic purification, ultrafiltration and other processes, and establish a stable, scalable and traceable production process.
3. Establish standard system at detection stage: Adopt qPCR quantitative detection, complete full set of methodological verification, and formulate Standard Operating Procedure (SOP) and original records meeting review requirements.
4. Advance preparation at registration stage: Complete HCD research data are required for both Class II and Class III medical device applications. Advance detection and verification can shorten the review cycle and reduce supplementary review risks.
5. Adhere to safety bottom line at quality stage: Implement internal control standards higher than regulatory requirements, build market trust and brand value oriented to end-user safety.
Supplementary Reading
In the past two years, the medical aesthetics anti-aging track has undergone a profound transformation of underlying logic. From surgical facelift to hyaluronic acid physical filling, to the rise of "regeneration" and "repair" concepts, collagen injections have undoubtedly stood at the industry outlet.
According to incomplete statistics, as of March 2026, about 2,100 domestic medical device registration certificates related to collagen have been approved, including 104 Class III medical device certificates and 7 recombinant injectable collagen certificates. The three leading recombinant collagen enterprises represented by Jinbo Bio, Giant Bio and Chuangjian Medical all have relevant certifications.
| Registration Certificate No. | Registrant | Product Name |
| National Medical Device Approval 20213130488 | Shanxi Jinbo Biomedical Co., Ltd. | Recombinant Type III Humanized Collagen Lyophilized Fiber |
| National Medical Device Approval 20233131245 | Shanxi Jinbo Biomedical Co., Ltd. | Injectable Recombinant Type III Humanized Collagen Solution |
| National Medical Device Approval 20253130751 | Shanxi Jinbo Biomedical Co., Ltd. | Injectable Recombinant Type III Humanized Collagen Gel |
| National Medical Device Approval 20253132643 | Jiangsu Chuangjian Medical Technology Co., Ltd. | Cross-linked Recombinant Collagen Implant |
| National Medical Device Approval 20253132049 | Shaanxi Giant Biotechnology Co., Ltd. | Recombinant Type I α1 Subtype Collagen Lyophilized Fiber |
| National Medical Device Approval 20263130044 | Shaanxi Giant Biotechnology Co., Ltd. | Recombinant Type I α1 Subtype Collagen and Sodium Hyaluronate Compound Solution |
| National Medical Device Approval 20263130287 | Jiangsu Chuangjian Medical Technology Co., Ltd. | Recombinant Collagen-like Lyophilized Fiber |
Based on current detection data, officially approved leading brands perform well in HCD residue control. Products obtaining NMPA Class III medical device registration certificates must submit complete and detailed data including biological evaluation, among which strict HCD residue control verification is included.
In addition to the above enterprises, first-tier enterprises such as Juyuan Medical and Fu'erjia have also incorporated HCD detection into core quality control links. In the future, HCD detection will become a key quality control threshold for market access of the recombinant collagen industry.
Safety Product Selection Suggestions for Beauty Seekers
Take Class III medical device as the bottom line: Class III medical devices are under the highest risk control level, indicating that indicators such as HCD residue, cytotoxicity and sensitization have obtained official recognition.
Pay attention to the technical background of brands: Enterprises with technical accumulation in artificial biological valves, ECM materials and other fields have more mature capabilities in impurity removal and immunogenicity control.
Rationally view price and ingredients: Low-priced products often compress purification costs, and HCD control is the core safety cost. High-purity and low-impurity products correspond to higher production and quality control investment.
Collagen injection is not only a filling treatment, but also a "communication" with the human immune system. In this process, HCD detection acts as an invisible "safety lock" to block invisible risks. The medical aesthetics safety of beauty seekers comes not only from standardized physician operations, but also from the support of a strict whole-chain quality control system.